Plants of genus Echinacea were identified and described during the Lewis and Clark Expedition of 1804-1806. Commonly called coneflowers, Echinacea species, including the Eastern purple coneflower, are found wild only in North America and are probably native to New York. Other common names for purple coneflower include Sampson Root and Missouri Snakeroot.
Traditional medicinal use:
Herbal medicines derived from several species of the indigenous Echinacea genus were in use throughout the plains of North America long before the introduction of European medicines, primarily as treatments for various infectious diseases and wounds, including snakebite wounds and insect bites. Echinacea species were considered to be some of the most important types of medicinal herbs by the plains tribes. Aboriginal peoples on the North American prairies used this plant widely to treat pain (as an anesthetic), coughs and colds. The juice was used to bathe burns and added to the waters sprinkled over coals during the traditional purification ceremonies known as “sweats”.


Plant Characteristics:
Purple coneflowers will bloom for a couple months. Flowers stand up to 3′ tall. The flower center develops into a spiny, dome-shaped, orange or brown center “cone” as the seed head develops. Flower petals tend to droop down with age and include shades of pink and purple. Tapered leaves become smaller and thinner toward the top of the stem. Purple coneflower has a fibrous root system with short woody rhizomes. These native perennial plants are drought tolerant.
Current use:
While the flowers of Echinacea species are used to make an extremely popular herbal tea, it is more noteworthy that Echinacea is one of the most popular herbal medicines / dietary supplements in Europe and North America today. Most Echinacea supplements on the market are made from an extract of the roots, flower heads or both (Parsons et al., 2018). People choose to take echinacea supplements for an almost unending list of reasons. What’s known is that echinacea seems to activate chemicals in the body that stimulate the body’s immune system. What isn’t clear is whether or how health benefits are realized as a result of taking echinacea dietary supplements.
Figuring out how echinacea works is complicated. Echinacea is loaded with chemicals that can have a biological effect. One group of bioactive compounds in echinacea is caffeic acid derivatives (CADs), a group which includes echinacoside, cynarin, cichoric acid, caftaric acid and chlorogenic acids and which possesses antimicrobial and antioxidant activity. Alkylamides, a group of more than 30 lipophilic compounds shown to have high bioavailability and immunomodulatory effects, are another class of bioactive chemicals manufactured by the plant.
The best that modern medicine seems to be able to offer concerning whether echinacea works is a solid “maybe”. This isn’t very satisfying given that the effects of echinacea have been widely researched. Contradictory results from credible studies doesn’t deliver consumer confidence.
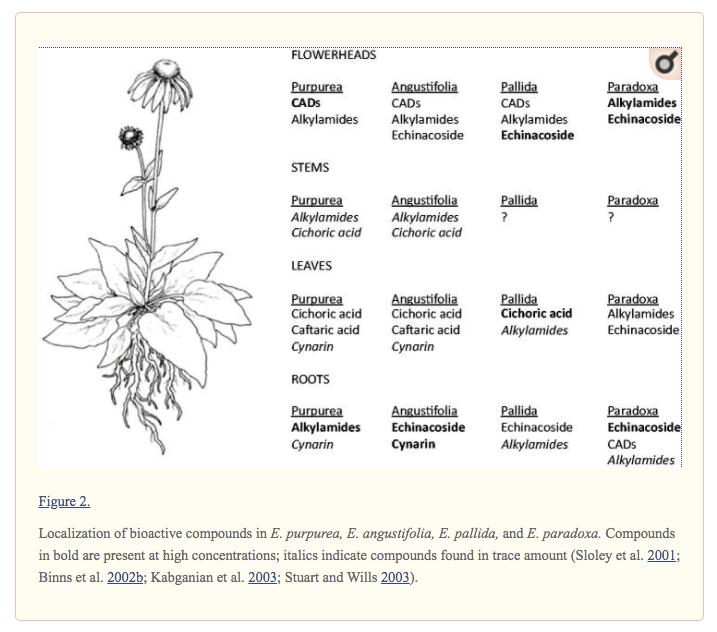
One reason that study results of echinacea effectiveness may legitimately differ is because the supplement itself is not standardized. Taking 400 mg of echinacea isn’t the same as taking 400 mg of aspirin because the composition and level of bioactive chemicals in 400 mg of echinacea can vary widely while the active ingredient in aspirin is always acetylsalicylic acid. The levels of various compounds in coneflowers can vary by genotype, plant parts, harvesting times and even by the level of stress to which a particular plant has been subjected. This variability in supplement source material is further complicated further by in variability post-harvest handling and processing. Given so many potential reasons for product irregularity, perhaps contradictory study results of echinacea effectiveness should be expected, not confounding.
The figure below, from Parsons et al., 2018, touches on the issue by showing how echinacea bioactive chemical distribution varies by genotype and plant part. The conclusion to draw is that the idea that someone can go buy a “standardized” echinacea supplement simply does not make sense at this time.
Stuff Almost Nobody Knows:
(for the perpetually curious)
If some bioactive chemicals produced by plants deliver health benefits to people, why wouldn’t those same compounds deliver health benefits to animals?
There are indications that echinacea can do just that. Studies have consistently found that Echinacea feed additives improve immune activity in many animals (Parsons et al., 2018). Mice whose regular long-term diet was supplemented with echinacea were found to live longer than those in a control group (Brousseau and Miller, 2005). Supplementing animal feed with echinacea appears to enhance the efficacy of certain vaccinations (Parsons et al., 2018). Purple coneflower added to the feed of undernourished cows and horses stimulated appetite and species in genus Echinacea are known to have been utilized by livestock for forage (Groen, 2005). American bison, which self-select what they eat, primarily eat grass but also consume minor amounts of herbs, shrubs, and twigs (Berini and Badgley, 2017). Taken together, these results and others suggest that growing Echinacea species for animal feed is a possible future market with considerable upside (Parsons et al., 2018). More work needs to be done to investigate the intersection of delivery method, dosage and benefit.
Echinacea grew in great profusion in the mixed grass prairie and open woods from southern Canada to Texas and east. While it is becoming a more popular garden plant, it is now uncommon to come across coneflowers in the wild; purple coneflower is listed as critically imperiled at the state level in Missouri. Humankind already dedicates considerably more land to raising plant-based food for animals than is dedicated to raising plant-based food for people. Systematically integrating Echinacea species and possibly other herbal plants into the growing routines already in place to produce animal feed could benefit animals, people and ecosystems.
Black Squirrel Farms strives for accuracy but everyone makes mistakes sometimes. We’re happy to update our information if needed. Please contact us if you spot an error or have a suggested edit, update or information inclusion. Help is welcome.
Black Squirrel Farms is not a provider of medical advice. We share information and ideas but do not make health recommendations for or against the use of any traditional medicinal plant for any reason.
References:
Aly, S. and Mohamed, M. Echinacea purpurea and Allium sativum as immunostimulants in fish culture using Nile tilapia (Oreochromis niloticus), https://pubmed.ncbi.nlm.nih.gov/20455962/, Journal of Animal Physiology and Animal Nutrition. 2010;94(5):e31–e39, 2010.
Berini, J. and Badgley, C., Diet segregation in American bison of Yellowstone National Park, https://blogs.biomedcentral.com/bmcseriesblog/2017/07/27/diet-segregation-american-bison-yellowstone-national-park/, BMC Series blog, 27 Jul 2017, 2017.
Brousseau M, Miller SC. Enhancement of natural killer cells and increased survival of aging mice fed daily Echinacea root extract from youth, https://pubmed.ncbi.nlm.nih.gov/16041619/, Biogerontology. 2005;6(3):157–163, 2005.
Groen, A., Echinacea angustifolia. In: Fire Effects Information System, https://www.fs.fed.us/database/feis/plants/forb/echang/all.html. U.S. Department of Agriculture, Forest Service, Rocky Mountain Research Station, Fire Sciences Laboratory, 2005.
Hudson, J., Applications of the Phytomedicine Echinacea purpurea (Purple Coneflower) in Infectious Diseases, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3205674/, J Biomed Biotechnol. 2012;2012:769896. doi:10.1155/2012/769896, 2012.
Parsons J., Cameron S., Harris C., and Smith, M., Echinacea biotechnology: advances, commercialization and future considerations, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6179083/, Pharm Biol. 2018;56(1):485-494. doi:10.1080/13880209.2018.1501583, 2018
